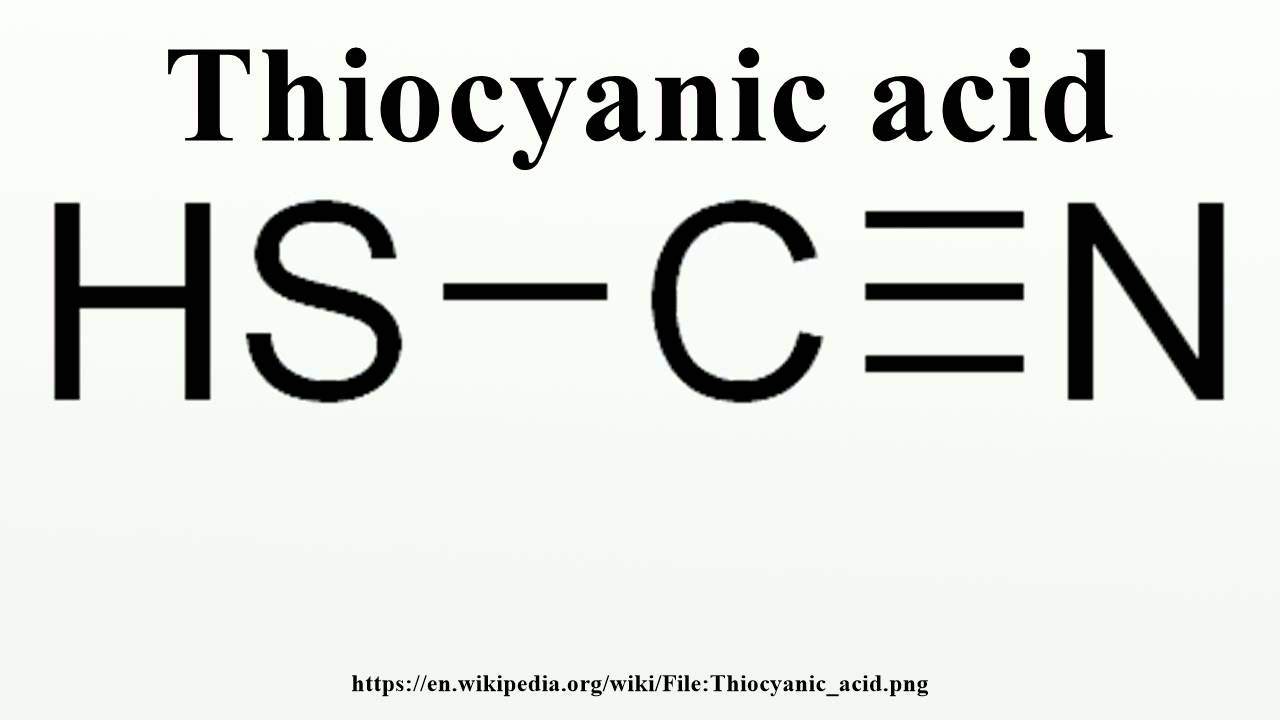Introduction
Thiocyanic acid is a chemical compound with the formula HSCN. It is a weak acid that is mainly known for its salts, called thiocyanates. The acid itself is a colorless liquid that is highly soluble in water and has a pungent odor.
Thiocyanic acid is mainly used in the production of thiocyanates, which are widely used in various industries. For example, potassium thiocyanate is used in pharmaceuticals, photography, and textile processing. Thiocyanates are also used as analytical reagents to detect the presence of metal ions.
In addition to its industrial applications, thiocyanic acid is found naturally in some foods, such as cabbage, broccoli, and mustard seeds. It is also a metabolic byproduct in humans and animals. However, its presence in high concentrations can be toxic.
Overall, thiocyanic acid plays an important role in various industries and has both natural and synthetic sources. Its properties and uses make it a significant compound to study and understand.
Chemical properties of Thiocyanic acid
Chemical properties of Thiocyanic acid include:
1. Acidic Nature: Thiocyanic acid is a weak acid and can undergo ionization in aqueous solutions, releasing hydrogen ions (H+). It can donate a proton to a base, forming thiocyanate ions (SCN-) and hydronium ions (H3O+). The dissociation reaction is as follows:
HSCN ⇌ SCN- + H+
2. Reactivity with Metals: Thiocyanic acid can react with certain metals, such as iron and copper, forming soluble thiocyanate complexes. For example, when iron reacts with thiocyanic acid, iron(III) thiocyanate (Fe(SCN)3) is formed.
3. Polymerization: Thiocyanic acid can undergo polymerization reactions, resulting in the formation of long-chain polymers known as thiocyanates. These polymers can have various physical and chemical properties, depending on the reaction conditions and the specific monomers involved.
4. Redox Reactions: Thiocyanic acid can participate in redox reactions, either as an oxidizing agent or reducing agent. For example, it can oxidize certain compounds by accepting electrons, while being reduced itself. The redox reactions involving thiocyanic acid can be used in analytical chemistry for the determination of various substances.
5. Reaction with Halogens: Thiocyanic acid can react with halogens, such as chlorine or bromine, forming respective halogen thiocyanates. These compounds are often used as intermediates in the synthesis of other chemicals or as reagents in certain reactions.
It is important to handle thiocyanic acid and its derivatives with caution, as they can be toxic and potentially harmful to health.
Production and uses of Thiocyanic acid
Thiocyanic acid, also known as sulfocyanic acid, is a chemical compound with the formula HSCN. It is a weak acid that is mainly produced by the reaction between hydrogen sulfide and hydrogen cyanide. Thiocyanic acid is colorless and has a pungent odor.
One of the most common uses of thiocyanic acid is as a precursor for the synthesis of various chemicals. It can be used in the production of pharmaceuticals, dyes, herbicides, and insecticides. It is also used as a reagent in organic chemistry for the preparation of synthetic compounds.
In addition to its use in chemical synthesis, thiocyanic acid is used as a component in some industrial processes. For example, it can be used in the production of galvanizing baths for the electrolytic deposition of zinc coatings on metal surfaces. It is also used in some mining processes as a extracting agent for certain metals.
Thiocyanic acid and its derivatives have also found applications in various fields. For instance, it can be used in medical diagnostics as a reagent for testing the presence of certain chemical compounds in biological samples. It is also used in the textile industry as a dye-fixing agent to ensure the color fastness of dyed fabrics.
However, it is important to note that thiocyanic acid is toxic and corrosive, and proper precautions should be taken when handling it.
Safety considerations and hazards of Thiocyanic acid
Safety considerations and hazards of Thiocyanic acid:
1. Corrosivity: Thiocyanic acid is corrosive and can cause severe burns when in contact with the skin or eyes. Inhalation of its vapors can also cause respiratory irritation.
2. Toxicity: Thiocyanic acid is toxic if ingested or if absorbed through the skin. It can cause nausea, vomiting, and diarrhea if swallowed, and can also affect the central nervous system, causing dizziness, confusion, and seizures.
3. Sensitization: Some individuals may develop an allergic reaction or sensitization to thiocyanic acid upon repeated or prolonged exposure. This can result in skin rashes, hives, or other allergic symptoms.
4. Combustibility: Thiocyanic acid is flammable and can ignite when exposed to heat or flames. It can release toxic fumes when burned, such as hydrogen cyanide.
5. Incompatibility: Thiocyanic acid is incompatible with strong oxidizing agents, strong acids, and bases. Mixing it with these substances can result in violent reactions or even explosions.
6. Environmental hazards: Thiocyanic acid can be harmful to aquatic life and may cause long-term adverse effects in the environment. It should be properly contained and disposed of to prevent contamination of water bodies or soil.
7. Storage and handling: Thiocyanic acid should be stored in well-ventilated, cool areas, away from sources of heat, sparks, or open flames. Proper personal protective equipment, such as gloves, goggles, and laboratory coats, should be worn when handling this chemical.
It is crucial to consult the material safety data sheet (MSDS) and follow proper handling procedures and safety guidelines when working with thiocyanic acid to minimize the risk of accidents or exposure.
Conclusion
In conclusion, thiocyanic acid is a chemical compound with the formula HSCN. It is a weak acid that can be found in various chemical processes and industrial applications. It is known for its characteristic odor and its ability to react with metals and other compounds. While it has some practical uses, it should be handled with caution due to its corrosive nature.
Topics related to Thiocyanic acid
Thiocyanic acid – YouTube
Thiocyanic acid – YouTube
How To Name Acids – The Fast & Easy Way! – YouTube
How To Name Acids – The Fast & Easy Way! – YouTube
Perthiocyanic Acid – Yellow Chem GOOD! – YouTube
Perthiocyanic Acid – Yellow Chem GOOD! – YouTube
Acids and Bases – Basic Introduction – Chemistry – YouTube
Acids and Bases – Basic Introduction – Chemistry – YouTube
Carboxylic Acids, Typical Acids and Esters | Organic Chemistry | Chemistry | FuseSchool – YouTube
Carboxylic Acids, Typical Acids and Esters | Organic Chemistry | Chemistry | FuseSchool – YouTube
Naming Acids In Chemistry – YouTube
Naming Acids In Chemistry – YouTube
Acidity: Crash Course Organic Chemistry #11 – YouTube
Acidity: Crash Course Organic Chemistry #11 – YouTube
What Makes Something Acidic? | Acids, Bases & Alkali's | Chemistry | FuseSchool – YouTube
What Makes Something Acidic? | Acids, Bases & Alkali's | Chemistry | FuseSchool – YouTube
Top Strongest Acids Ever – YouTube
Top Strongest Acids Ever – YouTube
Powerful Nitric Acid VS Lock | तेजाब की ताकत देखकर डर लगता है | Khel Khatam – YouTube
Powerful Nitric Acid VS Lock | तेजाब की ताकत देखकर डर लगता है | Khel Khatam – YouTube

Abigail Gutmann Doyle is a renowned Organic chemistry professor in Los Angeles. Her research focuses on the development of new chemical transformations in organic chemistry. She has won awards such as: Bayer Early Excellence in Science Award, Phi Lambda Upsilon National Fresenius Award, Presidential Early Career Award for Scientists and Engineers, BMS Unrestricted Grant in Synthetic Organic Chemistry.